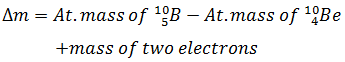Q.1.
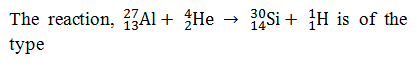
-
0%
nuclear fusion
-
0%
nuclear fission
-
0%
chemical reaction
-
0%
transmutation
Q.2.
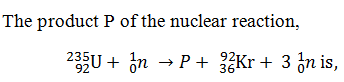
-
0%
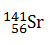
-
0%
2)
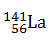
-
0%
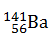
-
0%
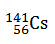
Q.3.
Consider the following nuclear reactions
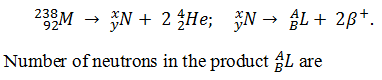
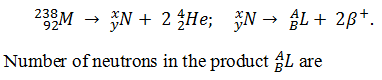
-
0%
142
-
0%
144
-
0%
140
-
0%
146
Q.4.
In which of the following nuclear reaction neutrons is emitted ?
-
0%
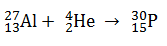
-
0%
2)
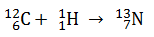
-
0%
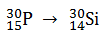
-
0%
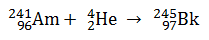
Q.5.
Match Column I and Column II and select the correct answer using the code given below the lists,
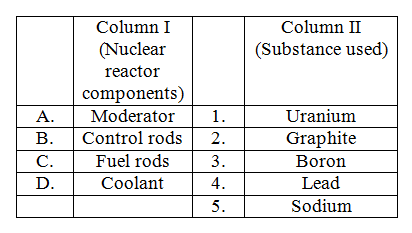

-
0%
A = 2, B = 1, C = 3, D = 5
-
0%
A = 2, B = 3, C = 1, D = 5
-
0%
A = 5, B = 2, C = 1, D = 3
-
0%
A = 3, B = 4, C = 1 , D = 2
-
0%
A = 4, B = 3, C = 2, D = 1
Q.6.
The age of a specimen t is related to the daughter/parent ratio of number of atoms (D/P) by the equation ( λ = decay constant)
-
0%
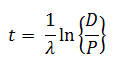
-
0%
2)
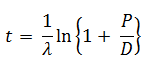
-
0%
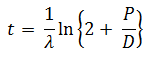
-
0%
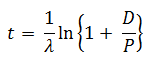
-
0%
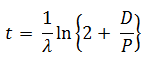
Q.7.
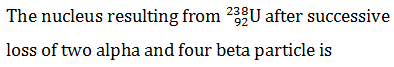
-
0%
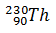
-
0%
2)
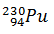
-
0%
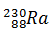
-
0%
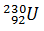
Q.8.

-
0%
II A
-
0%
IV B
-
0%
VI B
-
0%
VI A
Q.9.
which of the following isotopes occurs in nature?
-
0%
U233
-
0%
U230
-
0%
U226
-
0%
U238
Q.10.

-
0%
β– emission
-
0%
α– emission
-
0%
β+ emission
-
0%
K electron capture
Q.11.
The rate of disintegration of radioactive element changes from initial value of 10,000 dpm to 2,500 dpm in 50 days. The decay constant is
-
0%
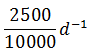
-
0%
2)
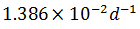
-
0%
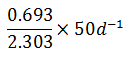
-
0%
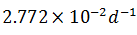
Q.12.
If 3/4 quantity of radioactive element disintegrates in two hours, its half-
life would be
-
0%
1 hour
-
0%
45 minutes
-
0%
30 minutes
-
0%
15 minutes
Q.13.
A radioactive isotope decays at such a rate that after 96 minutes, only 1/8 th of the original amount remains. The half-life of this nuclide in minutes is
-
0%
12
-
0%
24
-
0%
32
-
0%
48
Q.14.
What is the half life of a radioactive substance if 87.5 % of any given amount of the substance disintegrates in 40 minutes ?
-
0%
10 min
-
0%
20 min
-
0%
160 min
-
0%
13 min 20 sec
Q.15.
The half – life period of polonium is 140 days. If one starts with 1 g of polonium, 75% of it would have undergone distintegration in
-
0%
420 days
-
0%
280 days
-
0%
105 days
-
0%
35 days
Q.16.
The activity of a radioactive nuclide (Xis 6.023 curie at a certain time ‘t’. If its disintegration constant is 3.7 × 104 Sec−1 , the mass of X after t sec is
-
0%
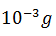
-
0%
2)
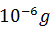
-
0%
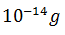
-
0%
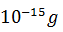
Q.17.

-
0%
3 neutrons
-
0%
2 – neutrons
-
0%
α − paticle
-
0%
β − paticle
Q.18.

-
0%
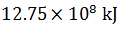
-
0%
2)
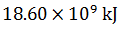
-
0%
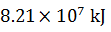
-
0%
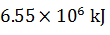
Q.19.
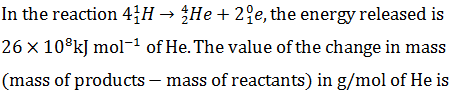
-
0%
0.029
-
0%
-0.029
-
0%
0.058
-
0%
-0.058
Q.20.
Wooden artifact and a freshly cut down tree give 7.6 and 15.2 counts min-1 g-1 of carbon (t1/2 = 5760 years) respectively. The age of the artifact is
-
0%
5760 years
-
0%
2)
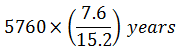
-
0%
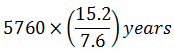
-
0%
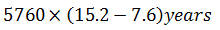
Q.21.
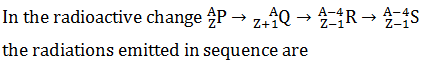
-
0%
α, β, γ
-
0%
β, α, γ
-
0%
γ, α, β
-
0%
β, γ, α
Q.22.
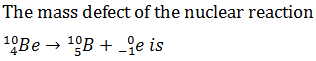
-
0%
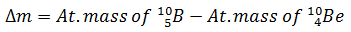
-
0%
2)
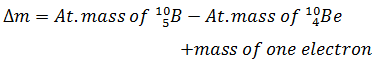
-
0%
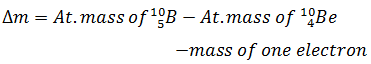
-
0%