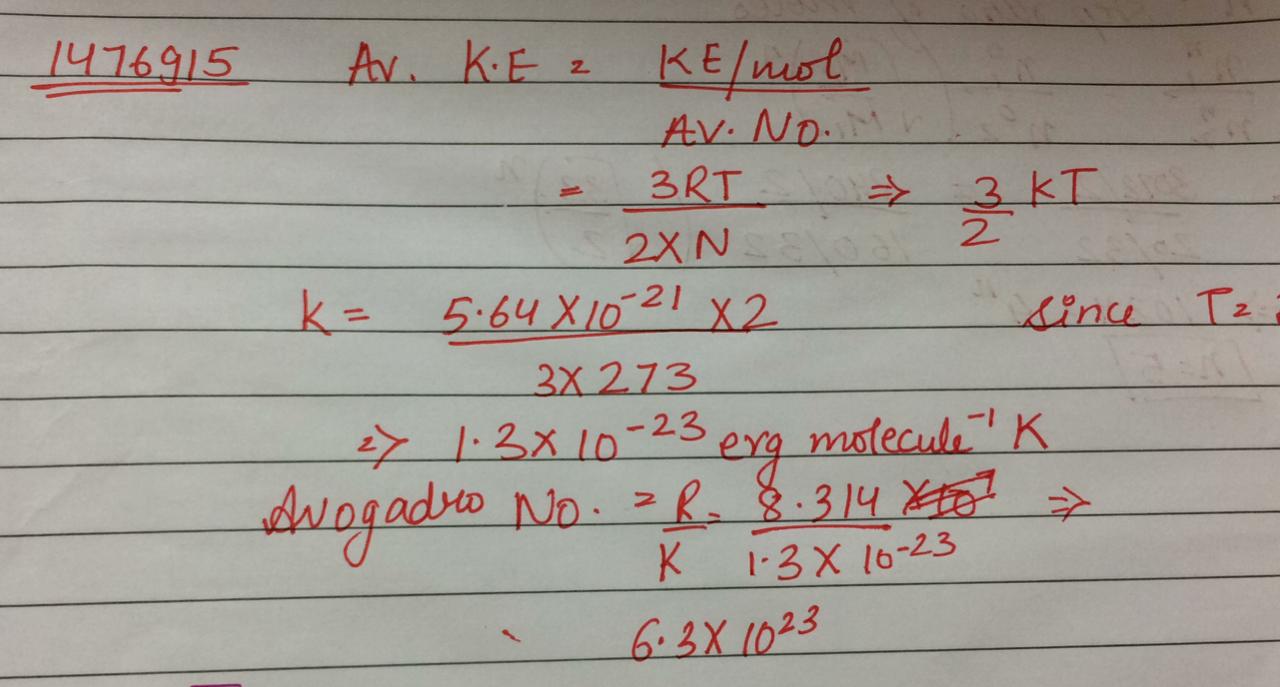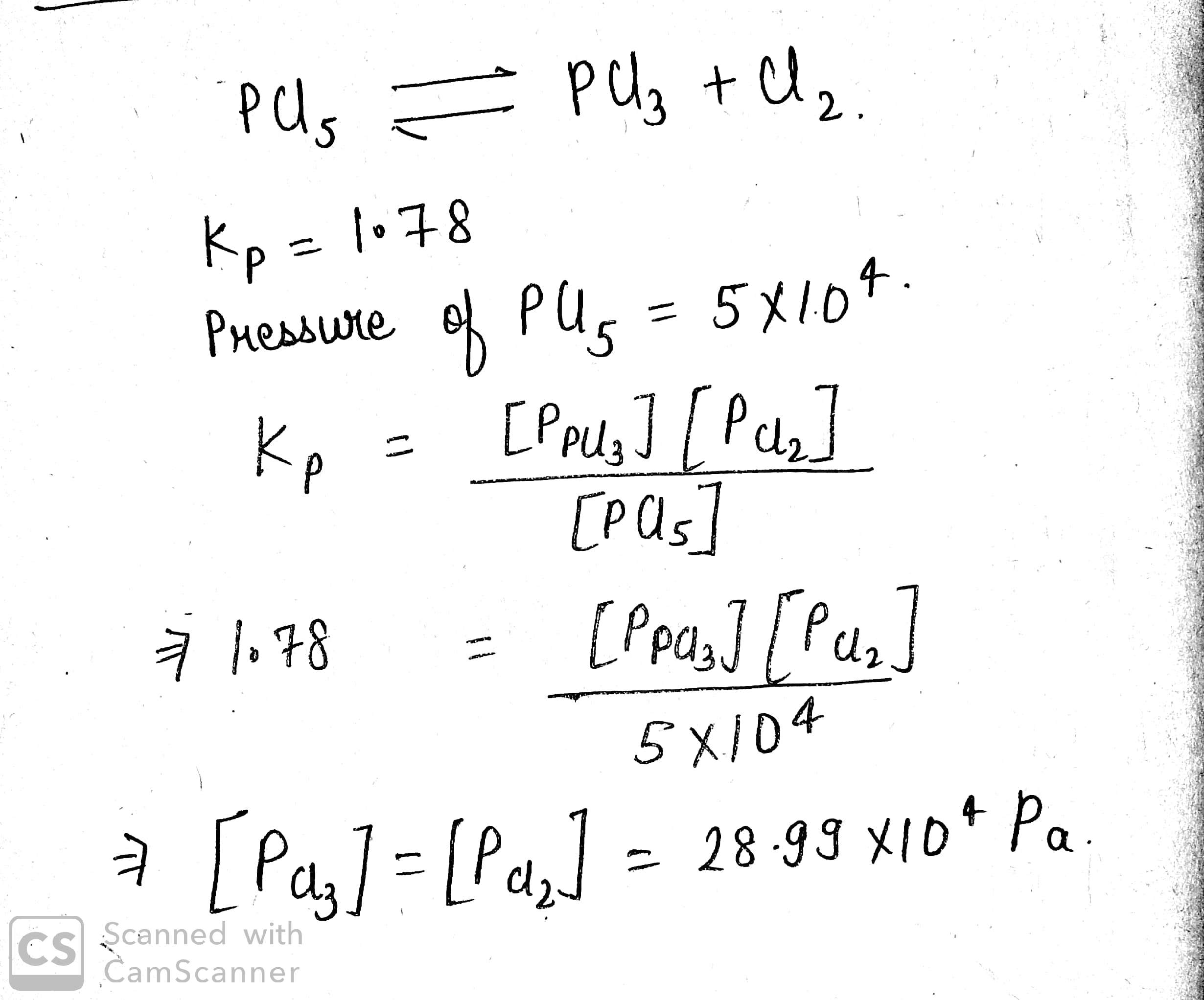States Of Matter Gases And Liquids - Class 11 Medical Chemistry - Extra Questions
Explain, why Dalton's law of partial pressure can be applied only to a mixture of non-reacting gases.
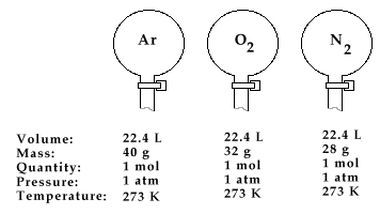
Equal volumes of all gases contain equal number of molecules under similar conditions of temperature and pressure.
If true enter 1, else enter 0.
One of the assumptions of kinetic theory of gases is that there is no force of attraction between the
molecules of a gas.
What are the assumptions of the kinetic molecular theory?
The volume occupied by the molecules of an ideal gas is :
Water vapours have more energy than water at same temperature.
Molarity of a substance ______ with increase in temperature.
The units of molarity is ______.
The $$P, \ V$$ measurements for $$100\ g$$ of phosgene gas at $$25^{\circ}C$$ are given below:
| $$P/ atm$$ | $$V/ dm^{3}$$ | $$PV/ atm\ dm^{3}$$ |
| $$0.1$$ | $$244.5$$ | $$24.45$$ |
| $$0.4$$ | $$61.02$$ | $$24.42$$ |
| $$2.0$$ | $$12.17$$ | $$24.34$$ |
| $$8.0$$ | $$2.925$$ | $$23.40$$ |
| $$9.8$$ | $$0.020$$ | $$0.20$$ |
| $$20.0$$ | $$0.020$$ | $$0.40$$ |
The vapour pressure of water at $$80^{\circ}C$$ is 355 torr. A 100 ml vessel contained water - saturated oxygen at $$80^{\circ}C$$, the total gas pressure being 730 torr. The contents of the vessel were pumped into 50.0 ml, vessel at the same temperature. What were partial pressures of oxygen and of vapour and the total pressure in the final equilibrium state ? Neglect the volume of any water might condense.
Write ideal gas equation for '$$n$$' moles of the gas.
Write any three postulates of Kinetic theory of gases.
What mass of hydrogen peroxide will be present in 2 litres of a 5 molar solution? Calculate the mass of oxygen which will be liberated by decomposition of 200 ml of this solution.
A closed bulb of capacity $$200$$ ml containing $$CH_4, H_2$$ and He at $$300$$K. The ratio of partial pressures of $$CH_4, H_2$$ and He, respectively is $$2 : 3 :5$$. Calculate the ratio of their weights present in the container.
Equal masses of $${ H }_{ 2 }$$, He and $${C H }_{ 4 }$$ are mixed in empty container at 300 K, when total pressure is 2.6 atm. The partial pressure of $${ H }_{ 2 }$$ in the mixture is:-
choose answer:
1) 0.5 atm
2) 1.22 atm
3) 0.8 atm
4) 0.2 atm
1) 0.5 atm
2) 1.22 atm
3) 0.8 atm
4) 0.2 atm
A straight glass tube has two inlets $$X$$ and $$Y$$ at the two ends. The length of the tube is $$200$$ cm. $$HCl$$ gas through inlet $$X$$ and $$NH_3$$ gas through inlet $$Y$$ are allowed to enter the tube at the same time. White fumes first appear at a point $$P$$ inside the tube. The distance of $$P$$ (in cm) from $$X$$ is:
Calculate the final volume (in L) of one mole of an ideal gas initially at $$0^o$$C and $$1$$ atm pressure, if it absorbs $$2000$$ cal of heat during a reversible isothermal expansion.
Set the following solutions in increasing order of vapour pressure:
$$3m$$ $$Na_2SO_4$$, $$1m$$ $$Urea$$ $$0.4m$$ $$AlCl_3$$$$0.2m$$ $$NaCl$$
An iron ball of radius $$0.3\ cm$$ falls through a column of oil of density $$0.94\ g\ cm^{-3}$$. If it attains a terminal velocity of $$0.54\ m\ s^{-1}$$, what is the viscosity of oil? Density of iron is $$7.8\ g\ cm^{-3}$$.
A viscous force of $$1.018\times 10^{-7}\ N$$ acting on a raindrop makes it fall through air with a terminal velocity of $$1\ ms^{-1}$$. If the viscosity of air is $$0.018\times 10^{-3}\ PI$$, what is the radius of the raindrop ?
Aerosol cans carry a clear warning of the heating of the can. Why?
One mole of an ideal gas expands reversibly and adiabatically from a temperature of $${ 27 }^{ }C$$. If the work done during the process is 3 kJ, then final temperature of the gas is:$$\left( { C }_{ V }=20\quad J/K \right) $$
(a) 100 K (b) 150 K
(c) 195 K (d) 255 K
Certain amount of a gas occupies a volume of $$0.4 litre$$ at $$17^oC$$. To what temperature should it be heated so that its volume gets (a) doubled (b) reduced to half, pressure remaining constant.
The pressure in a bulb dropped from $$2000$$ to $$1500$$ mm of mercury in $$47$$ min when the contained oxygen leaked through a small hole. The bulb was then evacuated. A mixture of oxygen and another gas of molecular weight $$79$$ in the molar ratio of $$1:1$$ at a total pressure of $$4000$$ mm of mercury was introduced. Find the molar ratio of the two gases remaining in the bulb after a period of $$74$$ min.
State and explain Gay Lussac's law of combining volumes.
The volume occupied by $$3.5$$g of $$O_2$$ gas at $$27^o$$C & $$740$$ mm of Hg pressure$$[O=16]$$
What are the main postulates of kinetic molecular theory of gases?
400 mL of oxygen at $$27^oC$$ were cooled to - $$15^oC$$ without the change in pressure. Calculate the contraction in volume.
Calculate the volume occupied by $$8.8g$$ of $$ { CO }_{ 2 }$$ at $$31.{ 1 }^{ o }C$$ and 1 bar pressure. ($$R=0.083$$ bar $${ dm }^{ 3 }\quad { K }^{ -1 }{ mol }^{ -1 })$$
A mixture of gases have $$O_2$$ and $$N_2$$ at the ratio of 1 : 4, then what will be the ratio of no. of molecular in mixture of gases?
A certain mass of a gas occupies a volume of $$0.2 dm^3$$ at $$273K$$. Calculate the volume (in $$dm^3$$)of the gas if its absolute temperature is doubled at the same pressure. (Round off to 1 decimal places)
Solids have infinite free surfaces while liquids have only one upper free surface.
A spherical balloon of 21 cm diameter is to be filled with $$H_2$$ gas at STP. From a cylinder, containing the gas at 20 atm and $$17^oC$$. If the vol. of cylinder is $$2.82$$ L, what will be the no. of balloon that can be filled up by this pressure?
State and explain Gay Lussac's law.
At constant volume of $$0.0821$$ litres for an ideal gas if $$\dfrac {d}{dT}(PT)=300$$ at $$150K$$. The number of moles of that gas is___
A bulb of tree litre capacity filled with air is headed from $$27^{o}C$$ to $$t^{o}C$$. The air thus expelled measured $$1.45$$ litre at $$17^{o}C$$ considering the pressure to be $$1$$ atm, throughout the experiment and ignoring the expansion of bulb, calculate $$t$$.
Two flask $$A$$ and $$B$$ of equal volumes maintained at temperature $$300K$$ and $$700 K$$ contain equal mass of $$He(g)$$ and $${ N }_{ 3 }(g)$$ respectively. What is the ratio of total translational kinetic energy of gas in flask $$A$$ to the flask $$B$$?
A gas occupies $$100.0 mL$$ at $${ 50 }^{ o }C$$ and 1 atm pressure. The gas is cooled at constant pressure so that volume is reduced to $$50.0 mL$$. What is the final temperature of the gas?
$$250$$ml of nitrogen gas maintained at $$650$$mm pressure and $$380 $$ mL of oxygen gas maintained at $$650$$ mm pressure are out together in 1 L flask.If temperature is kept constant, what will be the final pressure of the mixture?
The volume expansivity of a gas under constant pressure is $$0.0037$$ or $$ \left( \dfrac { 1 }{ 273 } \right) $$. Calculate its volume at $$-100^oC$$ if its volume at $$100^oC$$ is $$685 cm^3$$ .
Write down all the postulates of kinetic molecular theory of gases.
If $$200mL$$ of $$N_2$$ at $$25^oC$$ and a pressure of $$250mm$$ are mixed with $$350mL$$ of $$O_2$$ at $$25^oC$$ and a pressure of $$300mm$$ so that, the volume of resulting mixture is $$300mL$$, what would be the final pressure of the mixture at $$25^oC$$?
At a constant temperature, 250 mL of argon at 760 mm pressure and 600 mL of nitrogen at 500 mm pressure are put together in a one litre flask. Calculate the final pressure.
Some faculty members of XYZ classes were trying to simplify a problem for students by mentioning data explicity about a gaseous mixture. Information given by them is as follow.
Faculty - 1 : Mixture consist of three gases A, B and C.
Faculty - 2 : Molar mass of B is twice of A & Molar mass of A is $$4$$ times of C.
Faculty 3 : The gas that is neither heaviest nor lightest is $$16\%$$ by mass.
Faculty - 4 : Molar ratio of heaviest to lightest gas is $$2 : 5$$
Based on the above information, answer the question that follows.
What would be the average molar mass of mixture.
A gas occipies $$700ml$$ at S.T.P. Find the volume occupied by the gas when its pressure is $$400mm$$ of Hg and its temperature $$15^oC$$
A balloon is filled with hydrogen at room temperature, it will burst if pressure exceeds $$0.2$$ bar. If at $$1$$ bar pressure the gas occupies $$2.27$$ L volume, upto what volume can the balloon be expanded?
A gas occupies 100mL at 50degree C at 1atm pressure.The gas is cooled at constant pressure so that volume is reduced to 50mL.What is the final temperature of the gas
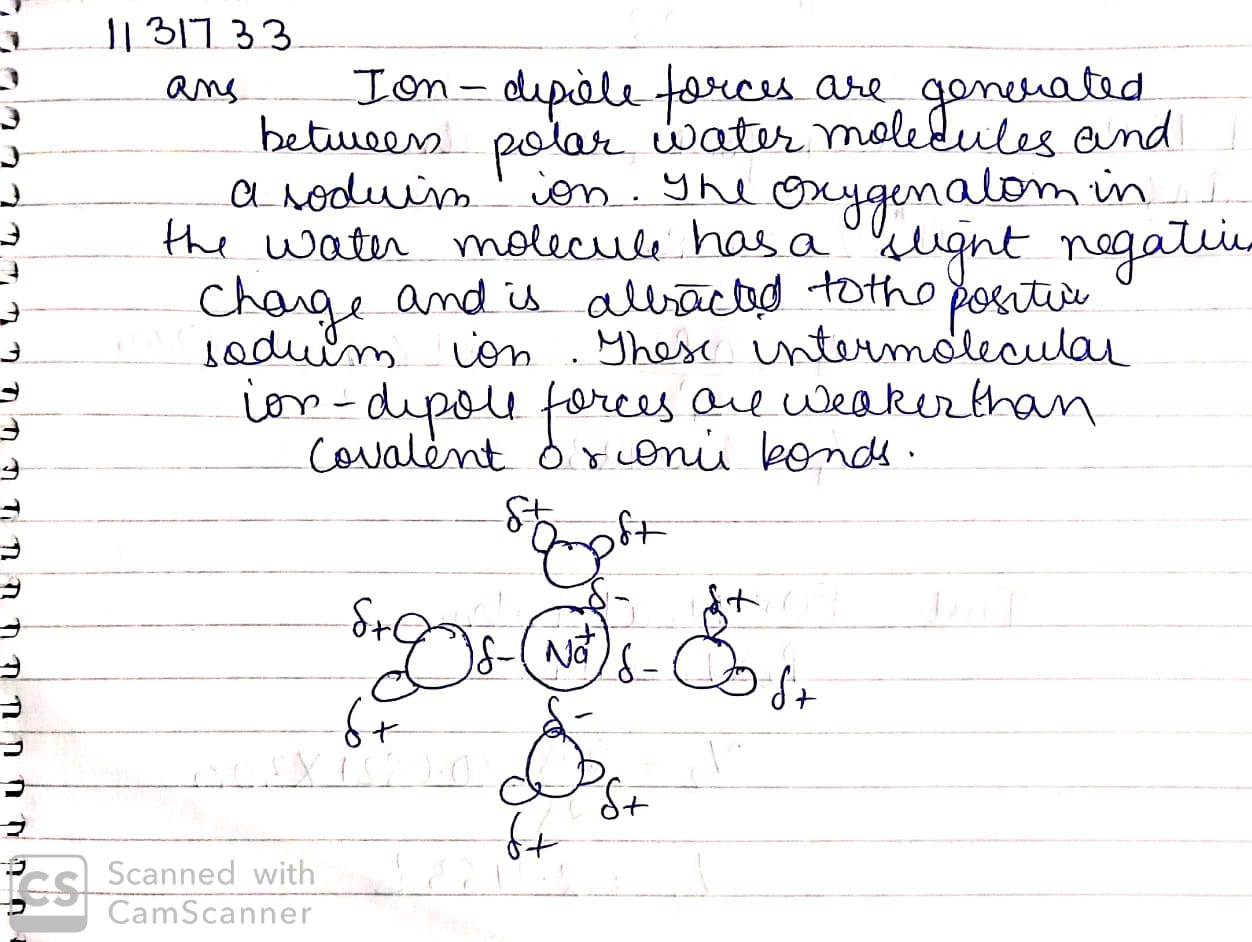
Explain ion-dipole interaction with suitable example.
Write note on Laws of multiple proportions.
Write note on Dalton's atomic theory.
The density of 3 M solution of NaCl is 1.25 g\ml. Calculate the molality of the solution.
A compound contains 4.07% hydrogen, 24.27% carbon and 71.65% chlorine. Its molar mass
is 98.96 g. What is its empirical and molecular formula?
Calculate number of atoms present in 500 ml 0.05 M aqueous solution of H$$_2$$SO$$_4$$?
Explain ion-dipole interaction with the help of suitable example.
The density of a gas is 3.80 g $$L^{-1}$$ at S.T.P calculate is density at $$27^{0}C$$ and 700 torr pressure.
$${O}_{2}$$ is present in $$1$$ litre flask at a pressure of $$1.52$$ x $${10}^{3}$$ $$mm$$ of Hg. Calculate mass of $${O}_{2}$$ at $$0C$$.
What percent of a sample of nitrogen must be allowed to escape if its temperature, pressure and volume are to be changed from $$220C$$, $$3 atm$$ and $$1.65 litre$$ to $$110C$$, $$0.7 atm$$ and $$1.00 litre$$ respectively?
What will be the volume of a given mass of a gas at a pressure 50 cm of Hg. If it occupies 250 ml at a pressure of 98 cm of Hg, keeping the temperature constant.
Two closed bulbs of equal volume (v) containing an ideal gas initially at pressure $${ P }_{ 4 }$$ and temperature $${ T }_{ 1 }$$ are connected through a narrow tube of negligible volume as shown in the figure below. The temperature of one of the bulbs is then raised to $${ T }_{ 2 }$$. What is the final pressure $${ P }_{ f }$$?

A $$5\ lit$$ flask containing $$N_{2}$$ at $$1$$ bar and $$25^{o}C$$ is connected to a $$4 lit$$ flask containing $$N_{2}$$ at $$2$$ bar and $$0^{o}C$$. After the gases are allowed to mix keeping both flasks at their original temperature, what will be the pressure and amount of $$N_{2}$$ in the $$5\ lit$$ flask assuming ideal gas behaviour.
Find out the numerical value of gas constant (R).
An open bulb containing air at $$19^0C$$ was cooled to a certain temperature at which the no. of moles of gaseous molecules increased by $$25$$%. What is the final temperature?
Calculate the percentage of free volume available in $$1$$ mole gaseous water at $$1$$ atm pressure and $$373\ K$$.
Calculate the volume occupied by $$7.0\ g$$ of nitrogen gas at $$27^{o}C$$ and $$750\ mm$$ $$Hg$$ pressure,
According to Henrys Law, in gases, an increase in pressure increase______.
5.40 gm of an unknown gas at $${ 27 }^{ \circ }C$$ Occupies the same volume as 0.14 gm hydrogen at $${ 17 }^{ \circ }C$$ and same pressure. The molecular weight of unknown gas is
Write the postulates of "kinetic theory of gases' .
Kinetic energy of oxygen molecule at$$ { 0 }^{ \circ }C$$ is $$5.64\times{10 }^{ -21 }J$$. Calculate the value of Avogadro's number. Given$$R=8.31J$$ mole$$^{-1}K^{-1}$$.
Write ideal gas equation for one mole of a gas.
At $$525$$K, the equilibrium constant of the reaction, $$PCl_5\rightleftharpoons PCl_3+Cl_2$$ is $$1.78$$ atm $$(K_p)$$. At what pressure should an equimolar mixture of $$Cl_2$$ and $$PCl_3$$ be taken for the pressure of $$PCl_5$$ to be $$5\times 10^4$$ Pa at equilibrium, volume remaining constant?
Give two characteristic properties of a liquid.
Match the graphs between the following variable with their names:
"One of the assumptions of the kinetic theory of gases is that there is no force of attraction between the molecules of a gas". State and explain the evidence that shows that the assumptions are not applicable for real gases.
Why do liquids flow ?
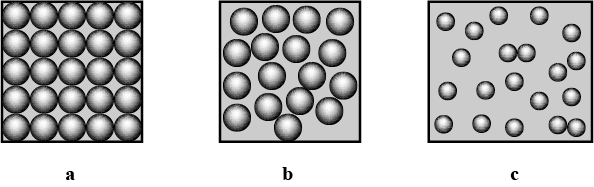
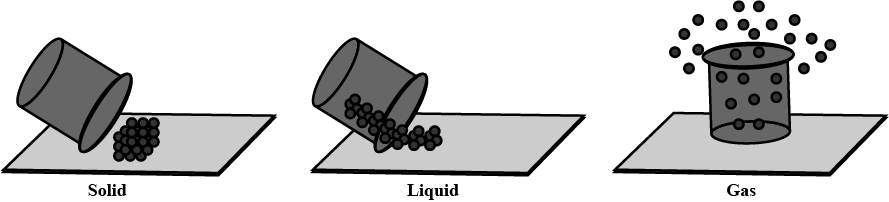
Define the three states of Matter
Define matter
For each of the following statements, say whether it describes a solid, a liquid, or a gas. (a) Particles move about very quickly.
(b) Particles are quite close together.
(c) Particles are far apart and move in all directions.
(b) Particles are quite close together.
(c) Particles are far apart and move in all directions.
Name the three states of matter.
Give the occurrence of water in the three different states i.e. solid, liquid and gaseous.
State three main characteristics of the particles of matter.
.......... deals with the study of matter and the changes it undergoes.
For the following statement, say whether it describes a solid, a liquid or a gas.
Particles are quite close together.
The different forms of a substance are called ............... of matter.
Give reason for : Tyres of automobiles are inflated to lesser pressure in summer than in winter.
Write a short note on Gay Lussac's Law.
Give two examples of substances that are rigid and not compressible.
State Dalton’s law of partial pressure.
What is Dalton's law of partial pressure?
What assumption can be made regarding the possibility of collision between gas molecules?
The drops of liquid take spherical shape. Why?
The boiling point of liquid changes on increasing the pressure. Why?
Mixture of $$NH_{3}$$ and $$HCl$$ gases do not follow Dalton's law of partial pressure. Why?
Complete the table
| Energy of gas molecules | Very high |
| Distance between the molecules | ......... |
| Freedom of moment of molecules | .......... |
| Attractive force between molecules | ........... |
State Ideal gas equation and law.
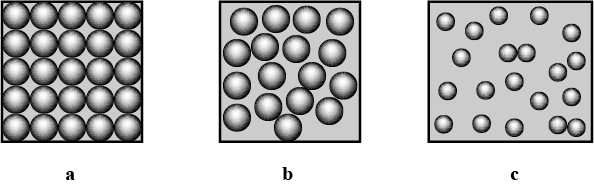
Identify and picturise the arrangement of particles in different states of matter.
Explain the peculiarities of materials.
At $$25^oC$$ and 760 mm of Hg pressure, a gas occupies 600 mL volume. What will be its pressure at a height where temperatures is $$10^oC$$ and volume of the gas is 640 mL?
Match the following.
| State of matter | A | B | |
| 1 | Gas | Soil | Milk |
| 2 | Solid | Kerosene | Oxygen |
| 3 | Liquid | Carbon Monoxide | Peas |
For hydrogen gas $$C_p-C_v=a$$ and for oxygen gas $$C_p-C_v=b$$,
so the relation between $$a$$ and $$b$$ is $$a = nb$$. Value of $$n$$ is:
so the relation between $$a$$ and $$b$$ is $$a = nb$$. Value of $$n$$ is:
Match the items of List 1 with those in List 2.
Match the following from $$Column-I$$ to $$Column-II$$.
300 litres of ammonia gas at $$20^0 C$$ and 20 atmosphere pressure are allowed to expand in a space of 600 litres capacity and to a pressure of one atmosphere .Calculate the drop in temperature.
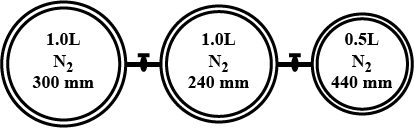
Consider the of bulbs shown arrangement below.
If the pressure of the system when all the stop cocks are opened is x (in atm) then find 100 x ? (760 mm = 1 atm)
$$16gm$$ of $${O}_{2}$$ was filled in a container of capacity $$8.21$$ lit. at $$300K$$. Calculate
(i) Pressure exerted by $${O}_{2}$$
(ii) Partial pressure of $${O}_{2}$$ and $${O}_{3}$$ if $$50$$% of oxygen is converted into ozone at same temperature
(iii) Total pressure exerted by gases if $$50$$% of oxygen is converted into ozone ($${O}_{3}$$) at temperature $$50K$$
Two flask of equal volume have been joined by a narrow tube of negligible volume. Initially both flasks are at $$300K$$ containing $$0.60$$ mole of $$O_2$$ gas at $$0.5atm$$ pressure. One of the flask is then placed in a thermostat at $$600K$$. Calculate final pressure and the number of $$O_2$$ gas in each flask.
$$20\ mL$$ of a solution containing $$0.2\ g$$ of impure sample of $$H_{2}O_{2}$$ reacts with $$0.316\ g$$ of $$KMnO_{4}$$ (acidic). Calculate:
(i) Purity of $$H_{2}O_{2}$$.
(ii) Volume of dry $$O_{2}$$ evolved at $$27^{\circ}C$$ and $$750\ mm\ P$$.
There are n connected container having volume V, 2V, 3V, ......, nV separated by stopcock. All container have same moles of gas at same temperature. If pressure of first container is P, then final pressure when all stop cocks are opened is ?
A mixture in which the mole ration of $${H}_{ 2 }$$ and $${O}_{ 2 }$$ is 2:1 is used to prepare water by the reaction
$${ 2H }_{ 2 }\left( g \right) +{ O }_{ 2 }\left( g \right) \longrightarrow { 2H }_{ 2 }O\left( g \right)$$
The total pressure of the container is 0.8 atm at 20 before the reaction. Determine the final pressure at 120 after reaction assuming 80% yield of water.
The kinetic molecular theory attributes an average kinetic energy of $$3/2\ KT$$ to each particle. What rms speed would a mist particle of mass $$10^{12}\ gm$$ have at room temperture $$(27^0{\circ}C)$$ according to the kinetic molecular theory.
At $$817^0C, K_p$$ for the reaction between pure $$CO_2$$ and excess hot graphite to form $$2CO(g)$$ is 10 atm.
What is the analysis of the gases at equilibrium at $$817^0C$$ & a total pressure of 4.0 atm? What is the partial pressure of $$CO_2$$ at equilibrium?
Calculate the value of $$R$$ in S.I unit for one mole of a gas.
(Given $$P={10}^{5}N/{m}^{2}\,V=0.0227098{m}^{3},\,T=273.15K,\,n=1mole$$)
Pure $$PCl_5$$ is introduced into an evacuated chamber and it comes to equilibrium at $$250^oC$$ and $$2\ atm$$. The equilibrium contains $$40.7\%$$ chlorine by volume. Calculate the partial pressures of all the gases present at equilibrium.
A quantity of an ideal gas is collected in a graduated tube over the mercury in a barometer type arrangement. The volume of gas at $$20^o$$C is $$50$$ ml and the level of mercury is $$100$$ mm above the outside of the mercury level. The atmospheric pressure is $$750$$ mm. Volume of gas at STP is :(Take $$R=0.083$$ it. atm/K/mole).
Calculate the total pressure in a 10 litre cylinder which contains o.4g He,1.6g oxygen and 1.4g nitrogen at$${ 27 }^{ 0 }C$$. Also calculate the partial pressure of He gas in the cylinder. Assume ideal behavious for gases :
An open container of volume $$V$$ contains air at temperature $$27^oC$$ or $$300K$$. The container is heated to such a temperature so that amount of gas coming out is $$2/3$$ of
(a) amount of gas initially present in the container.
(b) amount of gas finally remaining in the container.
Find the temperature to which the container should be heated.
Find out the temperature at which vessel was heated.
A gaseous mixture containing equal masses of $$He$$ and $$CH_4$$ is a total pressure of $$2 \ atm$$. Calculate partial pressure of $$He$$.
A gaseous mixture containing equal amount of nitrogen and methane has total pressure $$7.86$$ mm Hg at $$300$$K. Calculate the partial pressure (in mm Hg) of methane in the mixture.
Two litres of an ideal gas at a pressure of $$10$$ atm expands isothermally to a final volume of $$10$$ litres against a constant external pressure of $$1$$ atm. What will be the work done if process is done reversibly?
An evacuated bulb of known volume is filled with $$H_2$$ gas at room temperature $$(30^oC)$$. The pressure of the gas in the bulb is 750 mm Hg. A portion of the gas is transferred to a different flask and found to occupy a volume of 50.0 mL at 1 atm pressure and at the same temperature. The pressure of the $$H_2$$ gas remaining in the original bulb drops to 600 mm Hg. What is the volume of the bulb assuming $$H_2$$ gas is an ideal gas ?
Match the description in Column I with graph provided Column II. For $$n$$ moles of ideal gas at temperature $$T$$.
If the pressure and absolute temperature of $$2$$ liter of carbon dioxide are doubled, the volume of carbon dioxide would become (in litres)?
The density of liquid nitrogen is $$0.807 \, g \, mL_{-3}$$. If a person acidently swallowed a $$.028mL$$ drop of liquid nitrogen. What volume of nitrogen gas would be evolved in their body at $$100.0kPa$$ and $$27^oC$$?
If the kinetic energy of an electron is increased $$9$$ times then the wavelength associated with it would become.
For a gas adsorbed on a particular adsorbent at
$${O^ \circ }C,$$ the plot of $$\log \frac{x}{m}$$ versus log $$P$$ where $$P$$ is
in atm has a slope and intercept as shown in the fig.
Find the mass of the gas adsorbed by $$10$$ g of the adsorbent at $$0.2$$ atm.

1L flask containing vapour of methyl alcohol at a pressure of 1atm, and 25C was evacuated till the fill pressure was $${ 10 }^{ -3 }$$ mm.How many molecules of methyl alcohol were left in the flask?
At one bar pressure, the volume of a gas is 0.6 liter. If the gas receives 122 Joules of heat at one atmosphere pressure, the volume becomes 2 liters, then calculate its internal energy. (1 liter bar=101.32 Joule)
A mixture of helium and neon gases is collected over water at $${28.0^0}C$$ and $$745mm$$ of Hg, If the partial pressure of helium is $$368mm$$ of Hg, what is the partial pressure of neon? (Vapour pressure of water at $${28.0^0}C = 28.33$$ mm of Hg)
At $$400\ K$$ temperature in a closed vessel the $$\%$$ by volume of $$He,Ne$$ and $$Ar$$ are $$40\%, 40\%$$ and $$20\%$$ respectively. If the total pressure is $$25$$ bar, then find the partial pressure of each gas. (Total pressure is $$25$$ bar)
When the size of a soap bubble is increased by using more air in it, the surface area increases. Does it mean that the average separation between the surface molecules is increased?
All the substances are formed by small ............
What are the three states of matter ? Define each of them with two examples.
State the following
(c) Gas equation.
Give the assumptions of the kinetic molecular theory
Deduce the relation $$PV = nRT$$, where $$R$$ is a constant called universal gas constant.
How is the partial pressure of a gas in a mixture related to the total pressure of the gaseous mixture?
Explain the following observation : The tyre of an automobile is inflated at lesser pressure in summer than in winter.
Briefly explain Pressure-Temperature Law.
State and explain Daltons law of partial pressures. How is this law applied in the determination of the pressure of dry gas from that of the moist gas?
Class 11 Medical Chemistry Extra Questions
- Chemical Bonding And Molecular Structure Extra Questions
- Classification Of Elements And Periodicity In Properties Extra Questions
- Environmental Chemistry Extra Questions
- Equilibrium Extra Questions
- Hydrocarbons Extra Questions
- Hydrogen Extra Questions
- Organic Chemistry Some Basic Principles And Techniques Extra Questions
- Redox Reactions Extra Questions
- Some Basic Concepts Of Chemistry Extra Questions
- States Of Matter Gases And Liquids Extra Questions
- Structure Of Atom Extra Questions
- The P-Block Elements Extra Questions
- Thermodynamics Extra Questions
- The S-Block Elements Extra Questions

.bmp)